The difference between a thriving hydroponic operation and one that struggles often comes down to a few milliliters of solution and a tenth of a pH point. When Marcus Chen started his commercial lettuce operation three years ago, he thought nutrient management would be straightforward—just follow the manufacturer’s recommendations and watch the plants grow. Six months and several crop failures later, he learned that successful hydroponic nutrition requires understanding not just what nutrients plants need, but how they interact with water chemistry, environmental conditions, and each other in ways that can make or break a harvest.
Nutrient solution management represents the heart of hydroponic success. Unlike soil-based agriculture where plants can draw from a complex ecosystem of available nutrients, hydroponic plants depend entirely on what growers provide in their nutrient solutions. This complete dependency creates both opportunity and responsibility—the chance to deliver precisely what plants need for optimal growth, and the requirement to understand the intricate relationships between water chemistry, plant physiology, and environmental conditions.
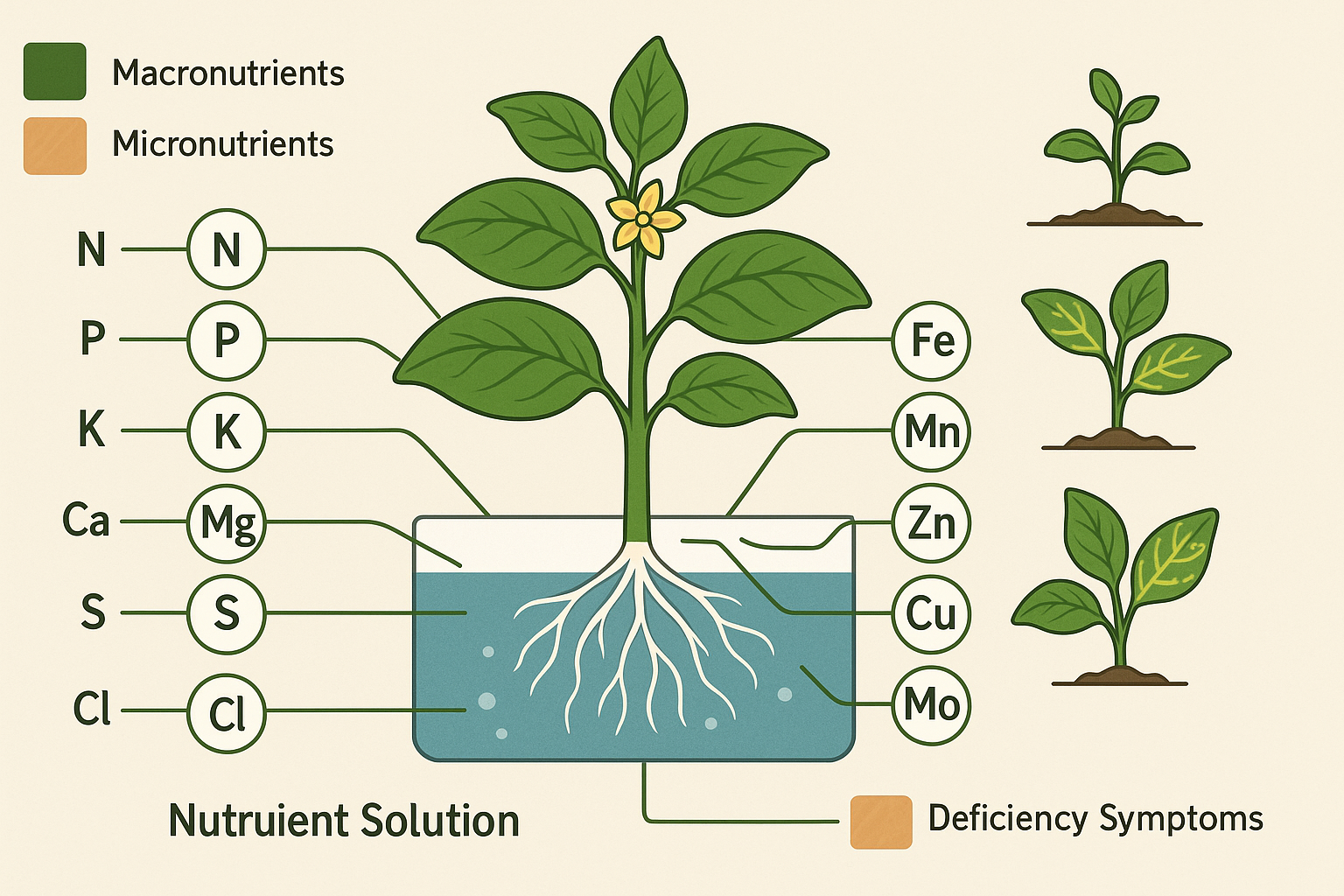
The Foundation: Understanding Essential Nutrients
Hydroponic solutions must supply all 16 essential nutrients that plants require for healthy growth and development. These nutrients fall into two categories: macronutrients needed in relatively large quantities and micronutrients required in trace amounts. The macronutrients include nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), and sulfur (S), while micronutrients encompass iron (Fe), manganese (Mn), zinc (Zn), copper (Cu), molybdenum (Mo), boron (B), and chlorine (Cl).
The challenge isn’t simply providing these nutrients—it’s delivering them in forms plants can readily absorb and in ratios that promote optimal growth without creating antagonistic interactions. Deficiency or excess of any nutrient can lead to physiological disorders that reduce yield, compromise quality, or kill plants entirely.
Nitrogen, the most abundant nutrient in most formulations, serves as the backbone of protein synthesis and chlorophyll production. Nitrate is the preferred nitrogen source for hydroponic systems due to its stability and efficient uptake characteristics. Unlike ammonium, which can acidify root zones and create toxicity at higher concentrations, nitrate provides consistent nutrition that plants can access readily across a wide pH range.
Phosphorus drives energy transfer and root development, particularly critical during seedling establishment and flowering phases. Potassium regulates water movement, enzyme activation, and stress resistance—factors that become increasingly important in controlled environment systems where plants may face rapid environmental changes.
Calcium deserves special attention in hydroponic systems. Beyond its structural role in cell walls, calcium affects membrane permeability and helps plants manage water stress. Calcium deficiencies manifest quickly in fast-growing hydroponic crops, causing problems like blossom end rot in tomatoes or tip burn in lettuce that can destroy entire harvests.
Water Quality: The Invisible Variable
Water quality fundamentally affects nutrient availability and uptake, making comprehensive water testing essential before formulating any nutrient program. The water source provides the foundation upon which all nutrient formulations are built, and its characteristics can either support or undermine even the most carefully designed nutrition programs.
Electrical conductivity (EC) measurements reveal the total dissolved solids in water, indicating both beneficial nutrients and potentially problematic salts. High EC source water may contain excessive sodium or chloride that can interfere with nutrient uptake or damage sensitive crops. Low EC water, while clean, may lack beneficial minerals like calcium and magnesium that plants need.
pH affects nutrient solubility and availability more than any other single factor. Optimal pH ranges between 5.5 and 6.5 for most hydroponic crops, providing the conditions where all essential nutrients remain soluble and accessible to plant roots. Outside this range, nutrients can precipitate out of solution or become chemically bound in forms plants cannot absorb.
Alkalinity, often confused with pH, measures the water’s buffering capacity—its resistance to pH changes. High alkalinity water requires more acid to adjust pH and tends to drift upward over time, requiring constant monitoring and adjustment. Understanding alkalinity helps predict how stable pH will remain and how much buffering acid the system will require.
Hardness, primarily calcium and magnesium content, affects both plant nutrition and system maintenance. Soft water may require calcium and magnesium supplementation, while very hard water can lead to mineral buildup in irrigation lines and equipment. Many successful growers find that moderate hardness (100-200 ppm CaCO3) provides beneficial minerals while avoiding excessive buildup issues.
pH and EC Management: The Dynamic Balance
Electrical conductivity levels indicate nutrient concentration and must be carefully managed to avoid plant stress while ensuring adequate nutrition. The relationship between EC, nutrient concentration, and plant uptake creates a dynamic system that requires ongoing attention and adjustment.
Optimal EC levels vary significantly by crop type, growth stage, and environmental conditions. Leafy greens typically thrive at EC levels between 1.2 and 1.8, while fruiting crops like tomatoes may require 2.0-3.0 EC during peak production. Seedlings need much lower concentrations (0.8-1.2 EC) to avoid salt stress during their sensitive establishment phase.
The relationship between pH and nutrient availability follows predictable patterns that experienced growers learn to anticipate. Iron becomes unavailable above pH 6.5, leading to chlorosis in new growth. Phosphorus precipitates with calcium at high pH levels, creating cloudy solutions and nutrient deficiencies. Manganese and zinc become less available as pH rises, while molybdenum availability increases.
pH naturally drifts in hydroponic systems as plants absorb nutrients selectively and respiration adds CO2 to the solution. Young, rapidly growing plants typically cause pH to rise as they absorb more cations than anions. Mature plants or those under stress may cause pH to drop as root respiration increases carbonic acid production.
Buffering systems help stabilize pH, but they must be balanced carefully to avoid interfering with nutrient uptake. Phosphoric acid serves as both a pH adjuster and phosphorus source, making it popular for hydroponic systems. Potassium hydroxide can raise pH while providing needed potassium, creating integrated pH and nutrition management.
Micronutrient Management: Small Amounts, Big Impact
Chelated micronutrients improve solubility and availability, preventing deficiencies that can severely impact crop quality and yield. While micronutrients represent less than 1% of most nutrient formulations by weight, their impact on plant health and productivity far exceeds their small concentrations.
Iron chelation deserves particular attention because iron deficiency is among the most common micronutrient problems in hydroponic systems. EDTA-chelated iron works well in most conditions, but DTPA or EDDHA chelates provide better stability at higher pH levels. The choice of chelating agent can determine whether plants receive adequate iron or develop the characteristic yellowing of iron-deficient new growth.
Manganese and zinc often work synergistically, but they can also compete for uptake sites if present in unbalanced ratios. Zinc deficiency commonly appears as shortened internodes and small, distorted leaves, while manganese deficiency causes interveinal chlorosis in young leaves. Both problems can develop quickly in fast-growing hydroponic crops if micronutrient ratios aren’t properly balanced.
Boron plays critical roles in cell wall formation and calcium transport, making adequate boron essential for crops susceptible to calcium-related disorders. However, boron toxicity can develop at concentrations only slightly above optimal levels, requiring precise management to avoid problems.
Copper and molybdenum are needed in extremely small quantities, but deficiencies can severely impact plant metabolism. Copper deficiency affects enzyme function and can cause wilting despite adequate water supply. Molybdenum is essential for nitrogen metabolism, and deficiencies can create symptoms that mimic nitrogen shortage even when nitrate levels are adequate.
Crop-Specific Nutrition Strategies
Nutrient requirements vary significantly by species and growth stage, requiring specific formulations to maximize yield and quality. Understanding these specific needs allows growers to optimize their nutrition programs for each crop they produce.
Leafy greens represent many growers’ introduction to hydroponic production because they’re relatively forgiving and have straightforward nutritional requirements. Lettuce, spinach, and herbs typically perform well with balanced, moderate-concentration solutions. However, balanced macronutrient ratios remain critical even for seemingly simple crops, as imbalances can affect taste, texture, and shelf life.
Tomatoes require more complex nutrition management due to their long growing season and distinct growth phases. Young plants need higher nitrogen ratios to support vegetative growth, while fruiting plants require increased potassium and calcium to support fruit development and prevent disorders like blossom end rot. Specific tomato formulations can significantly impact both yield and fruit quality.
Fruiting crops generally require higher EC levels than leafy greens because they produce more biomass and have greater nutritional demands. However, excessive EC can reduce fruit size and increase susceptibility to calcium-related disorders, requiring careful balance between adequate nutrition and osmotic stress prevention.
Herbs often have unique nutritional requirements that affect their essential oil production and flavor characteristics. Basil, for example, benefits from moderate nitrogen levels that promote leaf growth without encouraging excessive vegetative growth that can dilute flavor compounds. Many culinary herbs perform better with slightly lower EC levels than typical leafy greens.
Organic Approaches: Sustainable Alternatives
While traditional hydroponic nutrition relies on synthetic fertilizers, organic nutrient solutions derived from agricultural waste can match or exceed chemical formulations in effectiveness. This approach appeals to growers seeking sustainable production methods while potentially reducing input costs.
Research has shown that organic solutions developed from materials like banana peels can produce yields and quality comparable to conventional nutrient formulations, particularly for leafy green crops. These organic approaches often provide more complete nutrition than simple NPK formulations because they include naturally occurring micronutrients and organic compounds that can enhance plant growth.
The challenge with organic hydroponic nutrition lies in consistency and predictability. Organic materials can vary in nutrient content depending on source, processing methods, and storage conditions. Creating reliable organic nutrient solutions requires careful analysis and standardization processes that many small growers find challenging to implement.
Compost teas and liquid organic fertilizers can supplement conventional hydroponic nutrition, providing beneficial organic compounds and microorganisms that may enhance nutrient uptake and plant health. However, these materials must be filtered carefully to prevent clogging irrigation systems and should be used as supplements rather than complete nutrition programs.
Environmental Integration: Beyond the Solution
Light, temperature, and humidity significantly affect nutrient uptake and must be considered when designing nutrition programs. The interaction between environmental conditions and nutrition creates a complex system where optimal nutrient formulations can change based on growing conditions.
High light levels increase photosynthesis and nutrient demand, requiring higher EC levels to support increased metabolic activity. Conversely, low light conditions reduce nutrient uptake and may require lower EC levels to prevent salt stress. Many growers adjust their nutrition programs seasonally to account for changing light conditions.
Temperature affects both nutrient solubility and plant uptake rates. Warm conditions generally increase nutrient uptake but can also increase water consumption, potentially concentrating nutrient solutions beyond optimal levels. Cold conditions slow nutrient uptake and may require more frequent solution changes to prevent nutrient imbalances from developing.
Humidity influences transpiration rates and, consequently, nutrient uptake patterns. High humidity reduces transpiration and can lead to calcium deficiency symptoms even when calcium levels in the solution are adequate. Low humidity increases transpiration and may require more concentrated solutions to deliver adequate nutrition.
Advanced Monitoring and Automation
Regular testing and automation systems enhance nutrient management precision, allowing growers to maintain optimal conditions while reducing labor requirements. Modern monitoring systems can track multiple parameters simultaneously and make automatic adjustments to maintain optimal growing conditions.
Continuous pH and EC monitoring provides real-time feedback about solution stability and plant uptake patterns. These systems can detect problems before they become visible in plant symptoms, allowing for preventive management rather than reactive problem-solving.
Automated dosing systems can maintain target EC and pH levels by adding concentrated nutrient solutions or pH adjusters as needed. These systems reduce labor requirements while providing more consistent nutrition than manual management methods.
Nutrient solution analysis should extend beyond pH and EC to include periodic testing of specific nutrient levels. Ion-specific electrodes or laboratory analysis can reveal developing imbalances before they impact plant growth, allowing for proactive formula adjustments.
Data logging systems that track nutrient consumption patterns, environmental conditions, and plant growth responses provide valuable information for optimizing nutrition programs over time. This data can reveal subtle relationships between nutrition and performance that aren’t obvious from casual observation.
Troubleshooting Common Problems
Precipitation in nutrient solutions usually indicates pH problems, incompatible nutrients, or excessive concentrations. Calcium and phosphorus commonly precipitate at high pH levels, creating cloudy solutions and nutrient deficiencies. Iron can precipitate if chelation is inadequate or if pH rises too high.
Nutrient lockout occurs when excessive levels of one nutrient prevent uptake of others, even when the blocked nutrients are present in adequate concentrations. High potassium levels can block magnesium uptake, while excessive phosphorus can interfere with iron and zinc availability.
Salt buildup in growing media or on plant roots indicates excessive EC levels or inadequate drainage and solution exchange. This problem is particularly common in recirculating systems where evaporation concentrates nutrients over time.
Uneven growth patterns often indicate inconsistent solution delivery or concentration within the growing system. Dead spots in circulation, clogged emitters, or stratification in reservoirs can create zones with inadequate nutrition.
Economic Considerations and ROI
Nutrient costs typically represent 15-25% of operating expenses in commercial hydroponic operations, making efficient formulation and management critical for profitability. However, the cost of poor nutrition—reduced yields, quality problems, or crop losses—far exceeds the savings from using inferior or inadequately managed nutrient programs.
Bulk purchasing of base nutrients and mixing custom formulations can reduce costs compared to pre-mixed solutions, but requires technical knowledge and quality control procedures to ensure consistent results. Many successful operations find that hybrid approaches—using commercial base formulations supplemented with custom additions—provide the best balance of convenience, cost control, and performance.
Waste reduction through recirculating systems and solution management can significantly impact nutrient costs. Systems that recapture and reuse drainage solution can reduce nutrient consumption by 30-50% compared to run-to-waste systems, though they require more sophisticated monitoring and management.
Yield and quality improvements from optimized nutrition programs often justify higher nutrient costs through increased revenue. Premium crops grown with carefully managed nutrition can command higher prices and suffer fewer post-harvest losses, improving overall profitability.
The Path to Mastery
Successful hydroponic nutrition management combines scientific understanding with practical experience and careful observation. The basic principles—providing all essential nutrients in available forms at appropriate concentrations and pH—remain constant, but their application varies with crops, systems, and growing conditions.
Starting with proven formulations and making incremental adjustments based on plant response and system performance provides a safer path to optimization than attempting to develop custom solutions immediately. Most commercial nutrient suppliers provide baseline formulations that work well for common crops, offering a foundation for learning and refinement.
Record keeping becomes essential for developing expertise and troubleshooting problems when they arise. Tracking formulations, adjustments, environmental conditions, and plant responses creates a database of experience that informs future decisions and helps identify patterns that might not be obvious without documentation.
The investment in understanding hydroponic nutrition pays dividends throughout a grower’s career. Plants that receive optimal nutrition grow faster, yield more, and produce higher quality crops while remaining more resistant to pests and diseases. For commercial growers, this translates directly to improved profitability and competitive advantage.
Mastering nutrient solution management requires ongoing attention to detail, continuous learning, and willingness to adjust practices based on results. The complexity can seem overwhelming initially, but the systematic approach of understanding individual components and their interactions makes the process manageable and ultimately rewarding. Success comes from recognizing that hydroponic nutrition is both an art and a science—requiring technical knowledge combined with careful observation and responsive management to achieve optimal results.