You check your pH meter and your heart sinks. Yesterday it was 7.0—perfect. Today it’s 6.2 and dropping. Your fish are showing signs of stress, your plants are struggling to uptake nutrients, and you’re watching weeks of careful system management unravel in real time. Welcome to the pH rollercoaster that separates successful aquaponics growers from those who give up in frustration.
As an intermediate aquaponics grower, you’ve moved beyond the basic “keep it between 6.0 and 7.0” advice. You understand that pH isn’t just a number to maintain—it’s the master control that determines nutrient availability, bacterial health, fish welfare, and plant productivity. But knowing it’s important and actually managing it successfully are two very different challenges.
The reality is that pH management in aquaponics is more complex than in traditional hydroponics or fish keeping because you’re balancing the needs of three different biological systems: fish, plants, and bacteria. Each has different pH preferences, and the bacterial processes that make aquaponics possible are constantly driving pH changes that work against system stability.
Most intermediate growers have experienced the frustration of pH swings that seem to come from nowhere. You test religiously, make careful adjustments, and still wake up to find your system has drifted into dangerous territory overnight. This isn’t failure—it’s the natural behavior of aquaponics systems that haven’t been properly understood and managed.
Mastering pH control requires understanding the underlying processes that drive pH changes, implementing proactive management strategies, and developing the diagnostic skills to prevent small problems from becoming system disasters. The difference between struggling with pH and mastering it lies in moving from reactive crisis management to proactive system stewardship.
The Hidden Forces: Understanding What Drives pH Drift
pH drift in aquaponics isn’t random—it’s the predictable result of biological processes that are essential for system function. Understanding these processes allows you to anticipate changes and manage them proactively rather than reacting to crises after they develop.
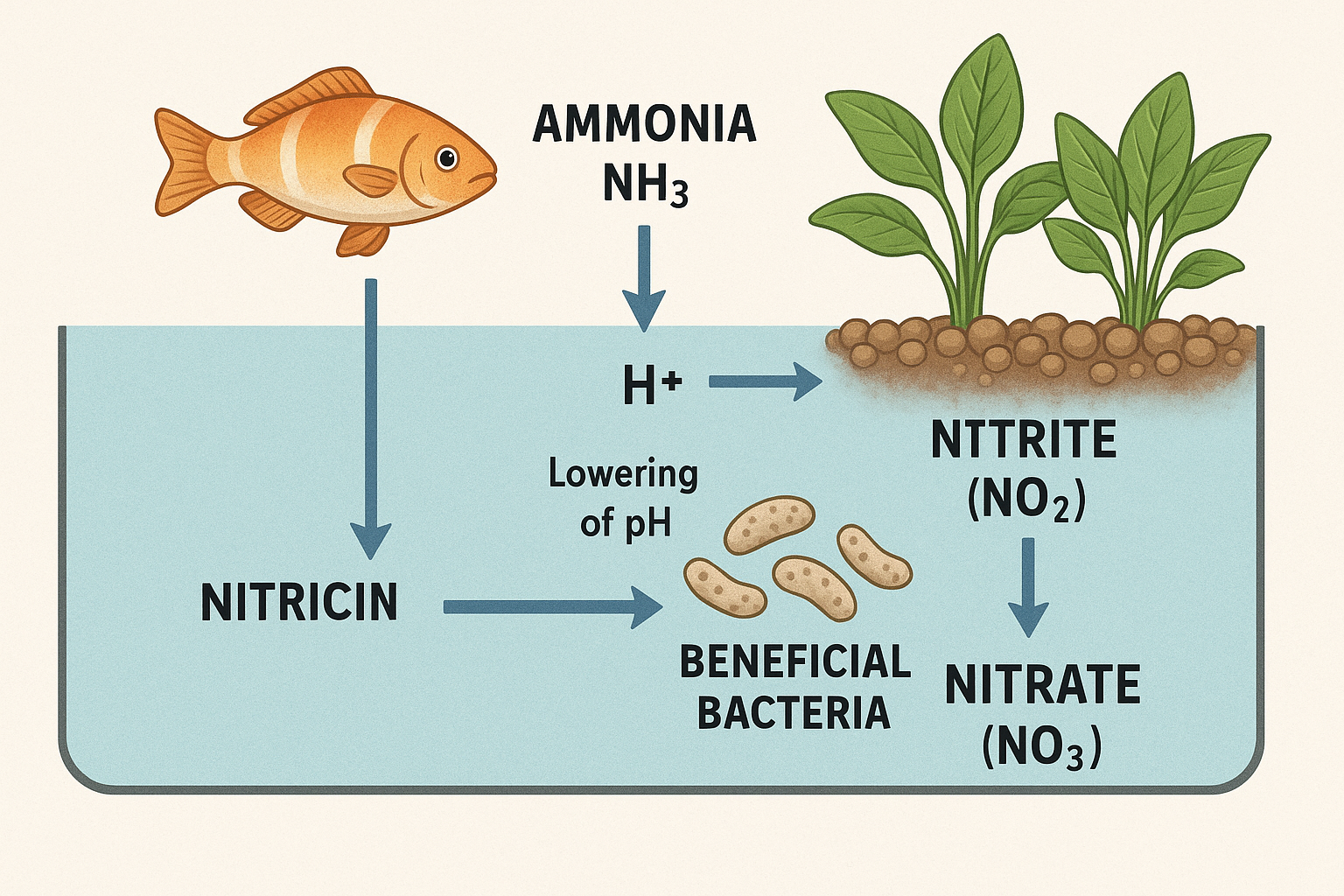
Nitrification: The Relentless pH Destroyer
Nitrification represents the most significant and constant downward pressure on pH in mature aquaponics systems. The nitrification process converts ammonia to nitrate while releasing hydrogen ions that lower pH. This isn’t a one-time event—it’s a continuous process that accelerates as your system’s bacterial populations mature and become more efficient.
The chemistry of nitrification reveals why pH problems intensify as systems mature. Each molecule of ammonia converted to nitrate releases two hydrogen ions into the water, directly lowering pH. As beneficial bacteria populations grow and become more efficient at processing fish waste, they actually accelerate the rate of pH decline.
Efficient biofilters, paradoxically, can create more severe pH challenges. Systems with highly developed biological filtration process ammonia more rapidly, potentially causing faster pH drops. This creates a situation where system improvement in one area (more efficient waste processing) creates challenges in another (pH stability).
The rate of nitrification—and thus pH decline—varies with temperature, oxygen levels, and bacterial health. Warmer temperatures accelerate bacterial activity, leading to faster pH drops. Higher oxygen levels support more robust bacterial populations, again accelerating pH decline. Understanding these relationships helps predict when pH challenges will be most severe.
Seasonal variations in nitrification rates create predictable patterns of pH stress. Summer months with warmer temperatures and higher biological activity often see accelerated pH decline. Winter periods with reduced bacterial activity may see slower pH changes but also reduced system efficiency.
The accumulation of nitrification over time means that pH problems compound. Small daily drops in pH, if not corrected, lead to increasingly acidic conditions that stress fish, inhibit plant nutrient uptake, and eventually harm the beneficial bacteria that drive system function.
Biological Activity: The Overfeeding Trap
Biological activity beyond nitrification creates additional pH challenges that many intermediate growers overlook. Overfeeding increases biological material in the system, causing ammonia spikes and corresponding pH drops. This creates a cascade of problems that extend far beyond simple waste accumulation.
Excess feed decomposes in the system, creating organic acids that directly lower pH while also providing substrates for bacterial growth that produces more hydrogen ions. This double impact can create rapid pH drops that are difficult to correct once they begin.
Dead plant material, fish waste, and unconsumed feed all contribute to biological loading that affects pH. Root debris, fallen leaves, and organic matter that accumulates in grow beds undergo decomposition processes that release organic acids and compete with beneficial bacteria for oxygen.
The timing of biological activity affects pH differently throughout the day. Bacterial activity peaks during warmer parts of the day, potentially creating diurnal pH swings that stress both fish and plants. Understanding these patterns helps time monitoring and adjustment activities for maximum effectiveness.
Bacterial populations that develop around excess organic matter may not be the beneficial nitrifying bacteria your system needs. Heterotrophic bacteria that consume organic matter can produce different waste products and create different pH effects than autotrophic nitrifying bacteria.
Denitrification: The Hidden pH Raiser
Denitrification occurs in anaerobic zones within the system, converting nitrate to nitrogen gas and potentially raising pH. This process often goes unnoticed because it happens in areas with poor water circulation—deep in grow bed media, in dead zones of tanks, or in areas where organic matter accumulates.
The pH-raising effect of denitrification can mask the pH-lowering effects of nitrification, creating false stability that suddenly disappears when anaerobic conditions change. This creates situations where pH seems stable for weeks, then suddenly drops precipitously when denitrification zones are disturbed.
Anaerobic zones in grow beds can create localized pH variations that affect plant root health even when overall system pH appears stable. Plants growing in areas with active denitrification may experience different pH conditions than those in well-oxygenated areas.
The balance between nitrification and denitrification in different parts of the system creates complex pH dynamics that require understanding of water flow patterns, oxygen distribution, and organic matter accumulation. Managing this balance requires attention to system design and maintenance practices.
System Materials: The Slow pH Influencers
System materials like growing media can cause gradual pH changes over time. These changes are often subtle and develop slowly, making them easy to overlook until they become significant problems.
Expanded clay pebbles, rock wool, and other growing media can slowly release minerals that affect pH. New media often has stronger effects than aged media, creating situations where systems experience gradual pH changes over the first few months of operation.
Tank materials, particularly concrete or cement-based components, can leach alkaline compounds that slowly raise pH over time. This effect is usually strongest when components are new but can continue at low levels for extended periods.
Plumbing materials and fittings can also contribute to pH changes, particularly if they contain metals that slowly corrode or plastics that release compounds over time. PVC pipes and fittings are generally pH-neutral, but some cheaper materials may have pH effects.
Water source characteristics interact with system materials to create unique pH challenges. Hard water with high mineral content may interact differently with system materials than soft water, creating site-specific pH management requirements.
Safe Buffering: The Art of pH Stabilization
Effective pH management in aquaponics requires buffering agents that stabilize pH without harming fish, plants, or beneficial bacteria. The choice of buffering agents and their application methods determine whether pH control enhances or disrupts system function.
Potassium Bicarbonate: The Grower’s Best Friend
Potassium bicarbonate raises pH and alkalinity while providing potassium for plant nutrition, making it safe for both fish and plants. This dual functionality makes it the preferred buffering agent for most aquaponics applications where pH needs to be raised.
The buffering mechanism of potassium bicarbonate works by neutralizing hydrogen ions while releasing potassium and bicarbonate ions into the water. The potassium provides essential plant nutrition, while the bicarbonate ions create alkalinity that resists future pH drops.
Dosage rates for potassium bicarbonate must be calculated carefully to avoid over-correction and potassium toxicity. Start with small amounts—typically 1-2 tablespoons per 100 gallons—and monitor pH response over several hours before adding more. The buffering effect develops gradually, so patience is essential.
Potassium bicarbonate dissolves readily in water, but it’s best added slowly to avoid shocking fish with rapid chemistry changes. Dissolve the buffer in a small amount of system water before adding it to the main circulation, allowing for gradual distribution throughout the system.
The alkalinity provided by potassium bicarbonate creates long-term pH stability by providing reserve buffering capacity. This means that systems treated with potassium bicarbonate resist pH drops for extended periods, reducing the frequency of pH adjustments needed.
Monitoring potassium levels becomes important with regular potassium bicarbonate use. While potassium is essential for plant growth, excessive levels can inhibit calcium and magnesium uptake, creating nutrient deficiencies despite adequate water levels of these minerals.
Calcium Carbonate: The Foundational Buffer
Calcium carbonate buffers pH drops while providing calcium for plant nutrition and supporting water hardness. This buffer works more slowly than potassium bicarbonate but provides longer-lasting pH stability.
The solubility characteristics of calcium carbonate make it ideal for providing sustained pH buffering. Unlike highly soluble buffers that provide immediate but short-term effects, calcium carbonate dissolves slowly, providing continuous pH support over extended periods.
Application methods for calcium carbonate vary depending on the desired buffering rate. Crushed oyster shell or limestone can be placed in grow beds or filter chambers to provide slow, continuous buffering. Powdered calcium carbonate can be mixed into water for faster effects when immediate pH correction is needed.
The calcium provided by calcium carbonate supports both plant nutrition and fish bone development. This dual benefit makes calcium carbonate particularly valuable in systems growing calcium-demanding crops like lettuce, spinach, and brassicas.
Water hardness effects from calcium carbonate can be beneficial in soft water regions where low hardness creates pH instability. The calcium and carbonate ions contribute to total hardness, providing chemical buffering that prevents rapid pH swings.
Combining calcium carbonate with potassium bicarbonate creates complementary buffering systems. The calcium carbonate provides baseline, long-term stability while potassium bicarbonate allows for fine-tuning and immediate adjustments.
Application Strategies: Timing and Technique
Successful buffering requires understanding not just which agents to use, but when and how to apply them for maximum effectiveness with minimum system disruption.
Gradual application prevents shocking fish and beneficial bacteria with rapid chemistry changes. Sudden pH swings, even in the “correct” direction, can stress system biology and create new problems. Small, frequent adjustments work better than large, infrequent ones.
Timing of buffer applications affects their effectiveness and system impact. Adding buffers during periods of high biological activity—typically during the day when plants are actively photosynthesizing—allows the system to accommodate changes more easily than during nighttime periods of reduced activity.
Distribution methods ensure even pH correction throughout the system. Adding buffers to areas with high water circulation helps distribute the effects evenly, while adding them to stagnant areas can create localized pH zones that stress plants or fish.
Monitoring response time helps predict future buffering needs. Some systems respond quickly to pH adjustments, while others change slowly. Understanding your system’s response characteristics helps time future adjustments and avoid over-correction.
Record-keeping of buffering activities helps develop system-specific management protocols. Tracking the amount of buffer used, the resulting pH change, and the duration of effect helps refine buffering strategies and predict future needs.

FarmHub tracks pH in real time and alerts you before a crash – not after. Start monitoring →
Testing Frequency: The Foundation of pH Control
Reliable pH management requires testing frequency that matches your system’s dynamics and stability. The right testing schedule provides early warning of problems while avoiding the expense and time commitment of excessive monitoring.
System Age and Testing Needs
Systems during start-up require daily testing to manage rapid pH changes as bacterial populations establish and biological processes stabilize. This intensive monitoring period is crucial for preventing catastrophic pH swings that can destroy beneficial bacteria and harm fish.
New systems experience rapid changes in bacterial populations, waste processing efficiency, and biological balance. These changes create unpredictable pH dynamics that require frequent monitoring to prevent problems. Daily testing during the first 6-8 weeks provides the data needed to understand your system’s specific pH characteristics.
The cycling process itself creates unique pH challenges as ammonia-oxidizing bacteria establish before nitrite-oxidizing bacteria, creating temporary imbalances that affect pH. Understanding these cycling dynamics helps predict pH changes and plan appropriate interventions.
Established systems typically require only weekly testing for routine pH monitoring, assuming no major changes in fish load, feeding rates, or environmental conditions. This reduced frequency reflects the greater stability of mature systems with established bacterial populations.
The transition from daily to weekly testing should be gradual, with testing frequency reduced as system stability improves. Consider reducing to every other day, then twice weekly, then weekly as pH stability demonstrates consistent patterns.
Seasonal changes may require temporary increases in testing frequency as temperature, daylight, and biological activity patterns change. Spring and fall transitions often create temporary pH instability that requires increased monitoring.
Critical Monitoring Periods
Certain system events and conditions require increased pH monitoring regardless of normal testing schedules. These critical periods represent times when pH problems are most likely to develop or when existing problems can quickly become severe.
Feeding changes—new feed types, increased feeding rates, or feeding schedule modifications—can alter pH dynamics within days. Any change in feeding practices should trigger increased pH monitoring for at least a week to detect problems early.
Water changes, particularly large ones, can disrupt pH stability by changing the chemical composition of system water. Monitor pH closely for several days after significant water changes to ensure stability returns.
Equipment failures or maintenance activities that disrupt water circulation, aeration, or biological filtration can create rapid pH changes. Test pH immediately after equipment problems and continue frequent testing until stability returns.
Weather events that affect system temperature, light levels, or humidity can indirectly affect pH by changing biological activity rates. Hot weather, power outages, or storms may require increased pH monitoring.
New plant additions or major harvests can change the biological balance of the system by altering nutrient uptake and root zone dynamics. Monitor pH more frequently for a week after major planting or harvesting activities.
Testing Equipment and Techniques
Reliable pH testing equipment is essential for accurate monitoring. The choice between digital meters, test strips, and liquid test kits affects both accuracy and convenience of pH management.
Digital pH meters provide the most accurate and precise measurements but require regular calibration and proper maintenance. Calibrate digital meters weekly with fresh buffer solutions, and store probes properly to maintain accuracy.
Liquid test kits offer good accuracy at lower cost but require careful technique and fresh reagents. Color comparisons can be difficult in artificial light, and reagent degradation affects accuracy over time.
Test strips provide convenient, quick results but with lower accuracy than other methods. They’re useful for rapid screening or backup testing but shouldn’t be relied upon for critical pH management decisions.
Testing location affects results, particularly in systems with uneven water circulation. Test pH at multiple locations initially to identify any variations, then establish consistent testing locations for comparable results.
Testing time of day can affect results due to diurnal variations in system biology. Establish consistent testing times—typically morning—to ensure comparable results and detect trends accurately.
Sample handling techniques affect testing accuracy. Use clean containers for water samples, avoid contamination from hands or equipment, and test samples promptly to prevent pH changes during storage.
Stability Techniques: Proactive pH Management
Maintaining stable pH requires more than responsive corrections—it demands proactive management strategies that prevent problems before they develop. These techniques focus on creating system conditions that naturally resist pH swings while maintaining optimal conditions for fish, plants, and bacteria.
Natural Buffering Systems
Natural buffering through carbonate-based systems and alkaline media provides sustainable pH stability. These approaches work with natural processes rather than against them, creating long-term stability with minimal ongoing intervention.
Crushed oyster shell placed in grow beds or filter chambers provides slow, continuous calcium carbonate buffering that prevents pH drops over extended periods. The shell dissolves gradually, providing sustained alkalinity without the need for regular buffer additions.
Limestone gravel or chips in biofilters serve similar functions to oyster shell while providing biological surface area for beneficial bacterial growth. The dual function of biological filtration and pH buffering makes limestone an efficient system component.
Coral sand or aragonite provide highly soluble calcium carbonate that responds quickly to pH drops. These materials work best in areas with good water circulation where they can dissolve and distribute effectively throughout the system.
Dolomite limestone provides both calcium and magnesium carbonate, creating broader buffering capacity while supplying two essential plant nutrients. The slower dissolution rate of dolomite provides long-term stability with minimal risk of over-correction.
Eggshells, thoroughly cleaned and crushed, provide an organic source of calcium carbonate that many growers find environmentally appealing. The dissolution rate is slower than commercial calcium carbonate, providing gentler buffering effects.
Organic Matter Integration
Organic matter like worm castings and compost teas offer natural pH buffering while providing plant nutrition. These approaches integrate pH management with overall system health improvement.
Worm castings added to grow beds provide gradual organic matter decomposition that creates natural buffering compounds. The humic and fulvic acids in worm castings help stabilize pH while improving nutrient availability for plants.
Compost teas introduce beneficial microorganisms that can help stabilize system biology and pH. The organic compounds in compost teas provide mild buffering while supporting beneficial bacterial populations.
Aged compost incorporated into growing media provides slow-release organic matter that creates natural buffering as it decomposes. The biological activity around decomposing organic matter can help stabilize local pH conditions around plant roots.
Biochar added to grow beds provides long-term pH buffering through its alkaline properties and high surface area for beneficial bacterial attachment. The porous structure of biochar creates microenvironments that can help moderate pH fluctuations.
Water Management Strategies
Strategic water changes help reset pH levels and provide temporary relief from pH stress. Effective water management goes beyond simple water changes to include source water treatment and timing strategies.
Partial water changes with properly pH-adjusted water can provide immediate relief from pH problems while diluting accumulated acids and waste products. Change 10-20% of system water when pH problems develop, using water adjusted to target pH levels.
Source water treatment addresses the root cause of some pH problems by removing or neutralizing compounds that contribute to pH instability. Reverse osmosis filtration removes dissolved carbonates and other minerals that can cause pH fluctuations, particularly in hard water areas.
Timing of water changes affects their pH impact. Adding water during periods of biological activity helps the system accommodate changes, while adding water during inactive periods can create temporary stress.
Water temperature matching prevents thermal shock while allowing for pH adjustment. Heat or cool replacement water to match system temperature, and adjust pH before adding to prevent additional stress.
Gradual water addition distributes the pH impact over time, preventing shock to fish and beneficial bacteria. Add replacement water slowly over several hours rather than all at once.
System Design Considerations
System design elements that support natural pH stability reduce the need for frequent interventions while creating more resilient systems overall.
Water volume affects pH stability by providing thermal mass and dilution capacity that moderates pH swings. Larger water volumes change pH more slowly than smaller volumes, providing more time for corrective action.
Biological diversity in the system creates more stable pH conditions by supporting varied bacterial populations with different pH effects. Diverse bacterial communities tend to create more stable overall conditions than monocultures.
Flow patterns that ensure good water circulation prevent the development of anaerobic zones that can create localized pH problems. Uniform water movement throughout the system helps maintain consistent pH conditions.
Aeration systems that maintain adequate dissolved oxygen levels support healthy bacterial populations that process waste efficiently without creating excessive pH swings. Proper aeration prevents the development of anaerobic conditions that can cause pH spikes.
Filtration systems that remove excess organic matter help prevent the biological loading that contributes to pH problems. Mechanical filtration removes particulates, while biological filtration processes dissolved waste products.
Advanced pH Management: Beyond the Basics
Intermediate growers ready to move beyond reactive pH management can implement advanced techniques that create more resilient, self-regulating systems. These approaches require more sophisticated understanding but provide superior long-term results.
Gradual Adjustment Protocols
Gentle pH adjustments using organic acids like citric acid provide careful pH lowering without shocking system biology. These protocols prioritize system stability over speed of correction.
Citric acid provides controlled pH reduction that’s particularly useful when pH rises above optimal levels. Use food-grade citric acid at very low concentrations—start with 1 gram per 100 gallons—and monitor pH response over several hours.
Acetic acid (vinegar) offers another organic option for careful pH reduction. Use only distilled white vinegar to avoid introducing oils or other compounds that might affect system biology. Start with very small amounts and increase gradually as needed.
Gradual adjustment protocols involve making small changes over extended periods rather than attempting rapid corrections. Plan pH adjustments over days or weeks rather than hours when possible, allowing system biology to adapt to changing conditions.
Multiple small additions work better than single large ones for most pH adjustments. Divide the total calculated adjustment into 4-6 smaller additions spread over 12-24 hours, monitoring pH response between additions.
Dilution techniques involve preparing pH-adjusted water separately and adding it gradually to the system. This approach allows for precise control over the final pH change while minimizing stress on system biology.
Monitoring Integration
Advanced pH management integrates pH monitoring with other water quality parameters to understand the complex interactions that drive pH changes.
Alkalinity testing provides crucial information about the system’s buffering capacity and help predict how pH will respond to various stresses. Test alkalinity monthly or whenever pH problems develop.
Nitrate monitoring helps predict pH trends by indicating the rate of nitrification occurring in the system. Rising nitrate levels often precede pH drops, providing early warning of developing problems.
Temperature correlation with pH changes helps predict seasonal pH challenges and optimize management strategies. Track pH alongside temperature to identify patterns and plan preventive measures.
Biological indicator monitoring—fish behavior, plant health, bacterial activity—provides early warning of pH stress before it becomes severe. Changes in these indicators often precede measurable pH changes.
Historical data analysis helps identify patterns and predict future pH challenges. Maintain records of pH changes, environmental conditions, and management interventions to develop system-specific management protocols.
Emergency Response Protocols
Even well-managed systems occasionally experience pH emergencies that require immediate intervention to prevent catastrophic losses.
Rapid response procedures for extreme pH swings prioritize life safety for fish while beginning gradual correction processes. Emergency protocols should be written, practiced, and immediately accessible during crises.
Fish protection measures during pH emergencies may include temporary relocation, emergency aeration, or rapid but controlled water changes. Have emergency equipment and procedures ready before problems develop.
System recovery protocols help restore normal pH management after emergency interventions. Recovery often takes longer than the initial correction and requires careful monitoring to prevent secondary problems.
Prevention strategies based on emergency experiences help avoid future crises. After each pH emergency, analyze the causes and implement preventive measures to reduce the likelihood of recurrence.
Mastering pH control in aquaponics requires patience, understanding, and consistent application of proven principles. The intermediate growers who succeed are those who move beyond crisis management to develop proactive systems that prevent problems before they develop. Your investment in understanding and managing pH will pay dividends in system stability, plant productivity, and fish health for years to come.
The path from struggling with pH to mastering it isn’t just about learning techniques—it’s about developing the systematic approach and diagnostic skills that separate successful growers from those who give up in frustration. Master pH control, and you’ll have taken the most important step toward becoming an expert aquaponics grower.
Stop checking pH manually. FarmHub connects your sensors, sets custom alert thresholds, and logs every reading automatically. Start free →